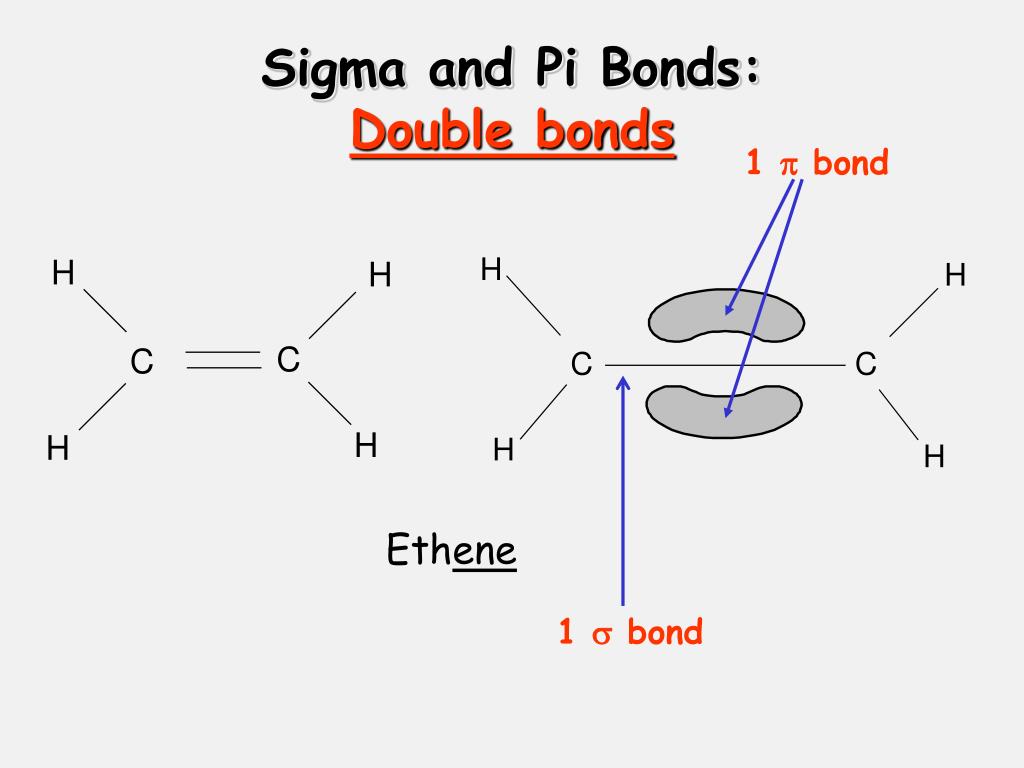 
Free rotation of atoms around pi bonds is not possible because it involves breaking the pi bonds. How many sigma and pi bonds are present in the following molecule a. Explanation: Lewis-dot structure: It shows the bonding between the atoms of a molecule as shown. The following example illustrates how a lone pair of electrons from carbon can be moved to make a new \(\pi\) bond to an adjacent carbon, and how the \(\pi\) electrons between carbon and oxygen can be. What is the formal charge on each atom in dichloromethane, CH 2Cl 2 a. The number of sigma and pi-bonds are eight and one respectively. The Lewis structures that result from moving electrons must be valid and must contain the same net charge as all the other resonance structures. There can be free rotation of atoms around the sigma bonds. electrons assigned to the atom in a Lewis structure. The total number of bonds formed by sulfur with two oxygen atoms is four. Every single bond is 1 sigma bond, every double. Among these, one is sigma bond and the second one is pi bond. To count sigma and pi bonds, draw the Lewis dot structure and count the single, double and triple bonds present. The bond angle between the hydrogen-carbon-hydrogen atoms is 109.5 and that in the carbon-nitrogen-oxygen atom is 120. Lewis structure of SCl 4 contains four S-Cl bonds. So, there will be two molecular geometry for separate entities of -CH3 and -NO2. framework -bond Overall structure Question: Identify the framework and. In other words, a single bond cannot be a pi bond. Hence each oxygen makes two bonds with sulfur atom. From the Lewis structure, we have got to know about two central atoms in a single nitromethane molecule. Thus, a pi bond is always present in molecules with multiple bonds, i.e., double or triple bonds. 
The reason is that the atoms constituting a single bond prefer to form a strong sigma bond rather than a weak pi bond. The reason is that the overlapping of atomic orbitals can take place to a greater extent during the formation of a sigma bond, whereas overlapping of orbitals occurs to a smaller extent during the formation of a pi bond.Ī pi bond between two atoms is formed only in addition to a sigma bond. The orbital overlap takes place in such a way that their axes are parallel to each other but perpendicular to the internuclear axis.Ī sigma bond is stronger than a pi bond. This type of covalent bond is formed by the lateral or sideways overlap of the atomic orbitals. Double and Triple bonds, however, contains sigma and pi bonds. If it is a single bond, it contains only sigma bond. The atomic orbitals overlap along the inter-nuclear axis and involve end-to-end or head-on overlap. Usually, all bonds between atoms in most organic compounds contain one sigma bond each. This type of covalent bond is formed by the axial overlapping of half-filled atomic orbitals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
